Technology
US Federal Funds Flowing: How Chipmakers Are Making The Most Of It

As the US continues to pour federal funds into various sectors of the economy, chipmakers are making the most of it. With billions of dollars in investments flowing into their coffers, chipmakers—and their suppliers—are continuing to invest in developing new technologies and production capacity that will remain competitive globally. In this blog post, we’ll explore how chipmakers are using these federal funds to grow their businesses and stay ahead of the competition. We’ll examine how they’re investing those funds, what benefits those investments provide, and how other companies can follow suit.
What are federal funds?
The federal funds rate is the interest rate at which depository institutions (banks and credit unions) lend reserve balances to other depository institutions overnight, on an uncollateralized basis. The federal funds rate is one of the most important prices in the U.S. economy, as it directly affects both monetary policy and borrowing costs.
In order to understand the federal funds rate, it’s necessary to first understand what reserve balances are. Reserve balances are deposits that banks keep at the Federal Reserve in order to meet their regulatory requirements. In other words, they’re like a savings account that banks use to cover any unexpected withdrawals or loan losses.
Banks are required to hold a certain percentage of their deposits as reserves, and they can choose to keep more than that if they want to. The amount of reserves a bank is required to hold depends on its size and the types of deposits it has. For example, large banks are required to hold more reserves than small banks.
When a bank has excess reserves, it can lend them out overnight to another bank that needs them. The interest rate that the lending bank charges is called the federal funds rate.
The federal funds rate is set by the market, which means that it’s determined by supply and demand for reserve balances. When there’s high demand for reserve balances (because many banks need them), the federal funds rate goes up. When there’s low demand for reserve balances (because few banks need them), the
How are chipmakers using federal funds?
The U.S. government has been awarding federal funds to chipmakers for many years now. In fact, it’s one of the main reasons why the United States has been able to maintain its leadership position in the global semiconductor industry.
Many of the world’s leading chipmakers, such as Intel, Qualcomm, and TSMC, have facilities in the United States and have received federal funds for research and development (R&D). These funds have helped these companies to develop new technologies and bring them to market quickly.
In addition to R&D, federal funds have also been used by chipmakers to build new manufacturing plants and expand existing ones. This has helped to create jobs and grow the economy.
So far, the Trump administration has proposed increasing federal funding for semiconductor R&D by $1 billion per year. This would be a big boost for the industry and would help to ensure that the United States remains at the forefront of this important technology.
What are the benefits of this for chipmakers?
As the world shifts to a more digital economy, the demand for semiconductors has exploded.
The US government has responded by investing billions of dollars in the semiconductor industry, with the goal of ensuring that American chipmakers can keep up with global demand.
This investment has already paid off, with American chipmakers reporting record sales and profits. The benefits of this government investment are twofold: first, it ensures that American chipmakers can compete in the global marketplace; and second, it helps to create high-paying jobs here at home.
It is clear that the US government’s investment in the semiconductor industry is paying off. Chipmakers are making the most of it by reporting record sales and profits. This is good news for both the economy and for American workers.
Are there any drawbacks?
Yes, there are some drawbacks to the US federal funds flowing into chipmakers. For one, it can be difficult to track where the money is going and how it’s being used. Additionally, some companies may not be using the funds in the most efficient way possible, which could lead to wastage. Finally, there is always the possibility that the government could change its mind about funding chipmakers, which could leave companies in a difficult position.
How will this impact the semiconductor industry in the long run?
The U.S. federal government is pouring money into the semiconductor industry, and chipmakers are poised to make the most of it.
The Trump administration has proposed a $1.3 billion investment in the semiconductor industry as part of its infrastructure plan. The funds would be used to support research and development, build new facilities, and create new jobs.
This is a welcome development for an industry that has been hit hard by falling demand and rising competition from China. The extra funding will help chipmakers maintain their competitive edge and continue to innovate.
In the long run, this could have a positive impact on the semiconductor industry as a whole. More investment in research and development will lead to more breakthroughs, which will benefit all players in the market. A strong U.S. semiconductor industry will also help to keep China in check, ensuring that it doesn’t become too dominant in the global market.
Conclusion
In conclusion, the US federal funds flowing into the chipmakers industries are a major boon for these businesses. With access to more resources and capital than ever before, chipmakers can make significant investments in their operations that will help them become even more competitive in the future. The extra funding will also increase research and development of innovative technologies as well as create new jobs that support local economies. Investing in chipmaker stocks is an attractive proposition for those looking to capitalize on this unique investment opportunity.
Equipments
Flood Light Online: Bright Outdoor Lighting Solutions

Outdoor lighting is essential for safety, security, and visibility in residential, commercial, and industrial environments. Among the many lighting solutions available today, flood lights remain one of the most effective options for illuminating large areas. Whether you need lighting for a parking lot, sports field, warehouse, garden, or building exterior, purchasing a Flood Light Online offers convenience and access to a wide variety of high-quality products.
Modern flood lights are designed to provide powerful illumination while maintaining energy efficiency. With advancements in LED technology, they have become more durable, cost-effective, and environmentally friendly than traditional lighting systems. Shopping for a Flood Light Online allows customers to compare products, evaluate features, and choose the best solution for their specific lighting needs.
What Is a Flood Light?
A flood light is a powerful lighting fixture designed to cast a broad beam of light across a large area. Unlike regular light fixtures that focus on smaller spaces, flood lights are built to deliver wide coverage, making them ideal for outdoor applications.
Flood lights are commonly used in residential properties, commercial facilities, sports venues, industrial sites, and public spaces. Their ability to provide bright and consistent illumination makes them a valuable addition to any location where visibility is important.
Why Buy a Flood Light Online?
Shopping for a Flood Light Online offers numerous benefits that traditional retail stores may not provide.
Greater Product Variety
Online stores typically carry a larger selection of flood lights than physical stores. Customers can browse different wattages, beam angles, color temperatures, and designs from various manufacturers.
Convenient Shopping
One of the biggest advantages of buying a Flood Light Online is convenience. You can browse products, compare specifications, and place an order from home without spending time visiting multiple stores.
Competitive Prices
Many online suppliers offer attractive discounts, promotional offers, and bulk pricing. This often results in better value compared to purchasing from local retailers.
Customer Reviews
Online marketplaces provide customer feedback and ratings that help buyers make informed purchasing decisions. Reviews often reveal valuable information about product quality, performance, and durability.
Easy Comparison
Comparing multiple flood lights online is simple. Customers can review specifications side by side and select the option that best matches their requirements and budget.
Common Applications of Flood Lights
Flood lights are used in a wide range of settings due to their versatility and powerful illumination capabilities.
Residential Lighting
Homeowners use flood lights for:
- Driveways
- Gardens
- Patios
- Garages
- Entryways
- Backyard spaces
Well-placed flood lights improve nighttime visibility and contribute to overall home security.
Commercial Properties
Businesses install flood lights to illuminate:
- Parking lots
- Shopping centers
- Building exteriors
- Outdoor work areas
- Loading docks
Proper lighting helps create a safer and more welcoming environment for customers and employees.
Industrial Facilities
Factories, warehouses, and manufacturing plants require reliable lighting for operational efficiency. Flood lights provide the bright illumination needed for outdoor workspaces and storage areas.
Sports Facilities
Flood lights are widely used in:
- Football fields
- Cricket grounds
- Tennis courts
- Basketball courts
- Athletic tracks
These lighting systems allow sporting activities to continue after sunset while maintaining excellent visibility.
Security Applications
Flood lights play a key role in security systems by reducing dark areas around buildings and discouraging unauthorized activity.
Features to Consider When Buying Online
Choosing the right flood light involves evaluating several important factors.
Brightness
Brightness is measured in lumens. The larger the area, the higher the lumen output required. Understanding the lighting needs of your space helps ensure optimal performance.
Wattage
Modern LED flood lights produce high brightness while consuming less power. Comparing lumen output rather than wattage alone provides a more accurate assessment of lighting efficiency.
Beam Angle
Beam angle determines how widely the light spreads.
- Narrow beam angles focus light on a specific target.
- Wide beam angles cover larger spaces.
Selecting the right beam angle ensures effective illumination.
Color Temperature
Color temperature influences the appearance of light.
- 2700K–3000K: Warm white
- 4000K–5000K: Neutral white
- 5000K–6500K: Cool white
Commercial and security applications often benefit from cooler color temperatures because they enhance visibility.
Weather Resistance
Outdoor flood lights should have a suitable IP rating to protect against rain, dust, and harsh environmental conditions.
Construction Quality
Look for fixtures made from durable materials such as aluminum with corrosion-resistant finishes. Quality construction helps ensure long-term performance.
Warranty
A strong warranty reflects manufacturer confidence and provides additional protection for buyers.
Tips for Selecting a Trusted Supplier
When purchasing a Flood Light Online, choosing a reliable supplier is essential.
Consider suppliers that offer:
- Detailed product specifications
- Clear warranty information
- Verified customer reviews
- Secure payment methods
- Responsive customer service
- Fast shipping options
A reputable supplier helps ensure a positive purchasing experience and reliable product performance.
Growing Demand for Online Purchases
As technology advances, more customers are choosing to buy lighting products online. Access to detailed product information, customer reviews, and competitive pricing makes online shopping an efficient solution for homeowners and businesses alike.
The increasing popularity of LED technology has also contributed to greater demand for flood lights that combine energy efficiency, durability, and high performance. Online stores continue to expand their product offerings to meet these evolving customer needs.
Conclusion
Purchasing a Flood Light Online is an excellent way to access reliable, energy-efficient, and high-performance lighting solutions for a variety of applications. Whether used for residential security, commercial properties, industrial facilities, or sports venues, flood lights provide powerful illumination that enhances visibility and safety.
By considering factors such as brightness, beam angle, color temperature, durability, and smart features, buyers can select the ideal flood light for their specific requirements. With the convenience of online shopping and the availability of advanced LED technology, finding the right flood lighting solution has never been easier.
Growth
Minding My Books: Expert QuickBooks Enterprise Solutions

Managing business finances effectively is one of the biggest challenges for growing companies in Canada. Tax rules, payroll requirements, and reporting standards can quickly become complex. As businesses expand, these responsibilities often become overwhelming without the right system in place. This is why choosing a reliable accounting solution—and an experienced consultant—matters more than ever. Minding My Books helps Canadian businesses simplify financial management through tailored QuickBooks Enterprise solutions.
Instead of offering generic setups, the company focuses on building systems that match each client’s unique business structure. From setup and migration to ongoing support, the goal is simple: make accounting easier, faster, and more accurate so business owners can focus on growth.
Smarter Approach to QuickBooks Enterprise Solutions
Minding My Books is a Canada-based consultancy that specializes in QuickBooks Enterprise setup, training, and long-term support. The firm understands that no two businesses operate in the same way. Because of this, it avoids one-size-fits-all configurations.
Instead, the process begins by studying how the business actually functions. The team looks at daily operations, financial workflows, and reporting needs. Then, they design a QuickBooks system that aligns with those real-world processes.
Whether the business is in retail, construction, manufacturing, professional services, or e-commerce, the setup is adjusted to fit its structure. This ensures that QuickBooks becomes a practical tool rather than a complicated system that slows teams down.
Customized Setup That Supports Business Needs
One of the biggest strengths of Minding My Books is its structured and personalized setup process. Every project starts with a detailed consultation. During this stage, the consultant identifies financial goals, operational challenges, and reporting expectations.
After that, the system is configured step by step. Key setup elements often include:
A well-structured chart of accounts designed around business operations
Accurate tax setup aligned with Canadian GST, HST, and provincial requirements
Payroll configuration to ensure compliance and timely employee payments
Custom financial reports that provide real-time insights into performance
This tailored approach ensures that business owners gain clear financial visibility from day one. It also reduces confusion and helps teams work more efficiently.
Seamless Integration Across Business Systems
Today’s businesses rely on multiple digital tools. Sales platforms, payment gateways, CRM systems, and inventory software all play an important role. However, managing data across disconnected systems often leads to errors and inefficiencies.
Minding My Books solves this problem by integrating QuickBooks Enterprise with other essential platforms. These may include Shopify, Stripe, Square, HubSpot, and inventory management systems.
As a result, businesses gain a connected ecosystem where accounting, sales, inventory, and reporting work together smoothly. This integration-first approach also supports long-term scalability, allowing businesses to grow without operational friction.
Industry-Focused Expertise
Every industry has different financial challenges. Minding My Books understands this clearly and adapts its solutions accordingly. For example, a construction company may need detailed job costing and project tracking. On the other hand, an e-commerce business may focus more on inventory management and sales tax compliance. Professional service firms may prioritize billing accuracy and expense tracking.
Because of this industry-focused approach, clients benefit in several ways:
- They streamline accounting workflows with greater efficiency
- They improve the accuracy of financial reports
- They stay compliant with Canadian tax and payroll regulations
This level of specialization ensures that QuickBooks is not just installed—it is optimized for real business use.
Ongoing Support Beyond Setup
Minding My Books does not stop after the initial configuration. Instead, it offers continuous support to ensure long-term success.
Businesses receive assistance with:
- Troubleshooting technical issues
- Data migration between systems
- Payroll processing and updates
- System optimization for performance
- Bookkeeping guidance and best practices
This ongoing support is especially valuable for growing businesses. As companies expand, their financial systems must evolve as well. Having a reliable partner ensures that QuickBooks continues to meet changing needs without disruption. Over time, many clients view Minding My Books as a long-term financial systems partner rather than just a service provider.
Making QuickBooks Enterprise More Powerful
QuickBooks Enterprise is already a powerful accounting platform. However, its true potential is unlocked only when it is configured correctly and aligned with business processes.
Minding My Books helps businesses go beyond basic usage. The focus is on transforming QuickBooks into a fully customized financial management system. This includes better reporting, improved automation, and stronger integration with other tools.
As a result, businesses gain:
- Better financial control
- Improved decision-making
- Reduced manual workload
- Higher accuracy in reporting
- Greater operational efficiency
This shift allows business owners to spend less time managing spreadsheets and more time focusing on strategy and growth.
Supporting Growth for Canadian Businesses
Canadian businesses face constant pressure to stay compliant while remaining competitive. With evolving tax regulations and increasing operational complexity, having the right accounting structure is essential.
Minding My Books supports this growth by offering solutions that scale with the business. Whether a company is just starting out or already operating at a larger scale, the systems are designed to grow alongside it. This flexibility ensures that businesses do not need to constantly rebuild their accounting setup as they expand. Instead, they can rely on a stable foundation that evolves with their needs.
Conclusion
Minding My Books stands out as a trusted QuickBooks Enterprise consultant for Canadian businesses that want clarity, efficiency, and long-term financial stability. Through customized setups, seamless integrations, industry-specific expertise, and ongoing support, the firm transforms QuickBooks into a powerful business tool.
Instead of struggling with complex accounting processes, businesses can focus on what truly matters—growth, performance, and decision-making. With the right guidance, financial management becomes not just easier, but also smarter and more strategic.
Consumer Services
White Label Services: A Smart Way for Agencies to Scale Faster

In today’s fast-moving digital market, businesses are always looking for smarter ways to grow without increasing operational pressure. This is where White Label services become a game-changer. Whether you run a startup, a marketing firm, or a growing agency, white labeling allows you to offer high-quality services under your own brand without building everything from scratch.
One of the fastest-growing segments in this industry is white label SEO services, as businesses increasingly seek expert SEO support without hiring full in-house teams. In this blog, we’ll explore how white label solutions work, their major benefits, and why businesses are increasingly choosing them to expand their service offerings.
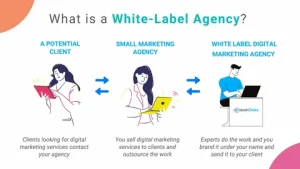
What Are White Label Services?
White label services are products or Solutions created by one company and rebranded by another company as their own. In simple words, a third-party provider does the work, while your business sells it under your brand name.
For example, a digital marketing company may not have an in-house SEO team. Instead, they can partner with a provider offering white label SEO services, deliver the service to clients under their own brand, and maintain full client ownership.
This business model has become highly popular because it allows businesses to focus on sales, client relationships, and growth while experts handle execution.
Businesses Choose White Label Solutions
Faster Business Growth
Building an internal team requires time, hiring costs, training, and management. White labeling removes these hurdles and allows agencies to expand services immediately.
A company can start offering:
- SEO
- PPC advertising
- Web development
- Content marketing
- Social media management
- Email marketing
without hiring separate specialists for each area.
Cost-Effective Operations
Hiring a complete in-house team can be expensive. Salaries, software, infrastructure, and employee management add significant costs.
A white label agency helps reduce:
- Recruitment costs
- Training expenses
- Operational overhead
- Software investments
This allows businesses to increase profits while maintaining service quality.
Access to Industry Experts
When businesses work with experienced white label partners, they gain access to professionals with specialized skills and updated industry knowledge. This means better results for clients without spending years building expertise internally.
How a White Label Agency Works
Step 1: Client Acquisition
Your business finds and communicates with clients. The customer only interacts with your brand.
Step 2: Project Outsourcing
Once the deal is finalized, the work is assigned to a trusted white label marketing agency.
Step 3: Service Delivery
The white label partner completes the project while following your brand guidelines.
Step 4: Branding and Reporting
Reports, dashboards, and deliverables are customized with your business branding before being shared with the client.
This process creates a seamless customer experience while your backend partner handles execution.
Popular White Label Marketing Services
SEO Services
SEO is one of the most in-demand white label marketing services today. Agencies often outsource:
- Keyword research
- Technical SEO
- Link building
- On-page optimization
- SEO audits
- Local SEO
Many businesses rely on white label SEO services to deliver measurable search engine rankings and organic traffic growth without building an internal SEO department.
PPC Advertising
Running paid ad campaigns requires expertise and constant monitoring. White labeling PPC management helps agencies deliver:
- Google Ads management
- Meta Ads campaigns
- Conversion optimization
- Ad copywriting
- ROI tracking
without hiring certified ad specialists.
Website Development
Many businesses outsource website projects to providers specializing in:
- WordPress development
- Shopify stores
- Custom websites
- Landing page design
- Website maintenance
This helps agencies take on more projects while maintaining delivery speed.
Social Media Management
Social media is essential for brand visibility. White label agency services often include:
- Content creation
- Scheduling posts
- Community management
- Paid social campaigns
- Performance reporting
This helps agencies serve clients consistently across multiple platforms.
Key Benefits of Marketing Services
Improved Scalability
Businesses can easily handle more clients without increasing internal workload.
When demand grows, the white label partner simply manages additional projects.
Stronger Brand Identity
Even though another company handles the work, all services are delivered under your branding. This strengthens your market position and builds client trust.
Better Client Retention
Offering multiple services under one roof makes clients less likely to switch providers.
When businesses provide SEO, PPC, web development, and social media together, customers prefer staying with a single agency.
Focus on Core Business Activities
Instead of managing delivery operations, agencies can focus on:
- Sales
- Marketing
- Customer relationships
- Business strategy
This improves efficiency and long-term growth.
Choosing the Right Label Partner
Check Industry Experience
Work with providers who have proven experience in your niche and understand current market trends.
Review Reporting and Communication
Transparent communication is important. A good partner should provide:
- Regular updates
- Branded reports
- Performance insights
- Quick response times
Evaluate Service Quality
Always review sample work, case studies, and client testimonials before partnering with a provider.
Ensure Scalability
Choose a partner capable of handling long-term growth as your client base expands.
Future of Label Services
The demand for outsourcing continues to grow as businesses look for flexible and scalable solutions.
Industries such as:
- Digital marketing
- Web development
- SaaS
- eCommerce
- Branding
- IT support
are rapidly adopting white label models to increase efficiency and expand globally.
As competition increases, businesses that adapt to scalable service models will have a stronger advantage in the market.
Conclusion
White labeling has become one of the smartest growth strategies for modern businesses. It allows companies to scale operations, increase revenue, and provide professional services without building large internal teams.
Whether you’re a startup or an established agency, partnering with the right provider can help you expand faster while maintaining strong branding and customer satisfaction. As the digital marketplace continues to evolve, businesses using strategic outsourcing models will remain more flexible, competitive, and profitable in the long run.
-
Business3 years ago
Cybersecurity Consulting Company SequelNet Provides Critical IT Support Services to Medical Billing Firm, Medical Optimum
-
Business3 years ago
Team Communication Software Transforms Operations at Finance Innovate
-
Business3 years ago
Project Management Tool Transforms Long Island Business
-
Business3 years ago
How Alleviate Poverty Utilized IPPBX’s All-in-One Solution to Transform Lives in New York City
-
health3 years ago
Breast Cancer: The Imperative Role of Mammograms in Screening and Early Detection
-
Sports3 years ago
Unstoppable Collaboration: D.C.’s Citi Open and Silicon Valley Classic Unite to Propel Women’s Tennis to New Heights
-
Art /Entertainment3 years ago
Embracing Renewal: Sizdabedar Celebrations Unite Iranians in New York’s Eisenhower Park
-
Finance3 years ago
The Benefits of Starting a Side Hustle for Financial Freedom