Development
ADC Manufacturing: Process, Challenges & Precision Oncology

The biopharmaceutical world is currently witnessing a historic shift. As of April 2026, Antibody-Drug Conjugates (ADCs) have evolved from experimental “magic bullets” into the cornerstone of precision oncology. These complex therapies work like biological cruise missiles, using antibodies to deliver lethal payloads directly to cancer cells while sparing healthy tissue.
However, the journey from a laboratory concept to a shelf-ready drug is incredibly steep. ADC manufacturing is a high-stakes discipline that blends advanced biology with volatile chemistry. For developers and procurement professionals, success in 2026 requires navigating a landscape defined by site-specific precision, extreme safety standards, and a rapidly expanding global market.
ADC Architecture: Antibody, Linker, and Payload
To understand the manufacturing challenge, one must first look at the three pillars that make an ADC functional. Each component requires a distinct expertise, and their integration is where the real complexity lies.
-
The Antibody (The GPS): This is the targeting system. Usually a monoclonal antibody (mAb), it is designed to seek out specific antigens on tumor surfaces. In 2026, manufacturers are prioritizing antibodies with faster internalization rates to ensure the payload enters the cell quickly.
-
The Payload (The Warhead): These are highly potent cytotoxic agents. While traditional microtubule inhibitors like MMAE are still used, the 2026 market has seen an explosion in Topoisomerase I inhibitors and even triple-payload constructs designed to overcome tumor resistance.
-
The Linker (The Fuse): The linker is the bridge. It must be stable enough to survive the blood’s circulation but smart enough to release the drug once inside the tumor. Modern linkers are often “cleavable,” triggered by specific enzymes or pH levels found only within the cancer cell.

How ADC Production Breaks Traditional Biologic Rules
Producing a standard antibody is difficult, but producing an ADC is significantly more dangerous and technically demanding. The primary difference lies in the Highly Potent Active Pharmaceutical Ingredients (HPAPIs).
Because the payloads are often 100 to 1,000 times more toxic than standard chemotherapy, the manufacturing environment must be a fortress. In 2026, facilities must operate under Occupational Exposure Band (OEB) 5 or 6 standards. This requires multi-level containment, specialized ventilation, and closed-loop systems to protect operators.
Furthermore, while traditional biologics focus on protein purity, ADC quality control centers on the Drug-to-Antibody Ratio (DAR). If the DAR is too low, the drug is ineffective; if it is too high, it becomes toxic to the patient. Achieving a consistent DAR is the “Holy Grail” of ADC production.
The 2026 Manufacturing Workflow
The production of an ADC is a synchronized dance between macromolecule biology and small-molecule chemistry.
1. Antibody Expression and Modification
Everything starts with the antibody. Typically grown in CHO (Chinese Hamster Ovary) cells within large-scale bioreactors, the antibody is purified using Protein A chromatography. In 2026, many developers now introduce “site-specific” modifications at this stage, creating precise “docking stations” on the antibody for the drug to attach to later.
2. Payload-Linker Synthesis
Simultaneously, the toxic payload and its chemical linker are synthesized in high-containment laboratories. This stage requires extreme chemical precision. The final payload-linker must be over 99% pure, as even trace impurities can drastically alter the drug’s safety profile.
3. The Conjugation Event
This is the most critical step. The antibody and the payload-linker are brought together in a controlled chemical reaction.
-
Historical Method: Random conjugation, which resulted in a “messy” mix of molecules.
-
2026 Standard: Site-specific conjugation. Using enzymatic or “click chemistry” methods, manufacturers can now ensure the drug attaches to the exact same spot on every antibody. This creates a highly homogeneous product that is easier for regulators like the FDA to approve.
4. Advanced Purification and Polishing
After conjugation, the mixture contains “free” toxic drugs that didn’t attach. These must be removed completely. Techniques like Hydrophobic Interaction Chromatography (HIC) and Tangential Flow Filtration (TFF) are used to “polish” the ADC, ensuring only the perfected conjugates remain.
5. Fill-Finish and Stabilization
Because ADCs are sensitive to light, temperature, and agitation, the final step involves stabilizing the drug. Many 2026 ADCs are lyophilized (freeze-dried) to extend their shelf life and ensure they remain potent until they reach the clinic.
Why the CDMO Model is Dominating in 2026
The sheer cost of building a facility that can handle both live cell cultures and deadly toxins is astronomical. As a result, the ADC Contract Development and Manufacturing Organization (CDMO) market is booming, estimated to reach over $2.1 billion this year.
By outsourcing to specialized partners, smaller biotech firms can access “end-to-end” solutions. These CDMOs provide the high-containment infrastructure and the analytical expertise (such as intact mass spectrometry) required to satisfy modern regulatory bodies. This collaborative model has significantly accelerated the timeline for new cancer therapies to move from the lab to the patient.
Regulatory Expectations and Quality Control
In 2026, the FDA and EMA have moved toward a more “holistic” review process. They no longer look at the antibody and drug separately; they evaluate the ADC as a single, integrated unit. Manufacturers must prove that their process is robust enough to produce the same DAR distribution every single time.
Key quality attributes now include:
-
Free Drug Levels: Must be nearly undetectable to prevent systemic side effects.
-
DAR Stability: Ensuring the drug doesn’t “fall off” the antibody while in the patient’s bloodstream.
-
Aggregation Prevention: Using surfactants to ensure the hydrophobic payloads don’t cause the antibodies to clump together.
Conclusion: The Path Forward
The evolution of ADC manufacturing represents one of the greatest technical achievements in modern medicine. By mastering the delicate balance of biology and chemistry, the industry is creating a new generation of “smart” medicines that are more effective and less toxic than ever before.
As we move further into 2026, the focus remains on making these processes faster, safer, and more consistent. For those who can navigate the complexities of site-specific conjugation and high-containment production, the reward is the ability to deliver life-saving precision to millions of patients worldwide. The era of the “magic bullet” has finally arrived, and it is being built one molecule at a time.
Development
Cambodia Visa Mistakes to Avoid & Guide for Cuban Citizens

If you are planning a trip to Cambodia, understanding Cambodia Visa Mistakes to Avoid and Cambodia Visa for Cuban Citizens is crucial for a smooth and successful travel experience. For official guidance and detailed support from the Cambodia visa company, you can visit Cambodia Visa Mistakes to Avoid and Cambodia Visa for Cuban Citizens, which provide complete instructions, eligibility details, and application requirements. These resources help travelers avoid errors and better understand the visa process before entering Cambodia.
Development
Precision Ophthalmic Surgical Instruments for Modern Eye Surgery
When performing delicate eye procedures, medical professionals require tools that offer absolute control. The margin for error in eye surgery is essentially non-existent, meaning surgeons need absolute reliability from their equipment. You need access to the best ophthalmic surgical instruments to ensure optimal patient outcomes and seamless surgical workflows.
The Complexities of the Human Eye
The human eye presents one of the most complex anatomical environments for any operating theatre. Surgeons navigate microscopic structures that demand unparalleled dexterity, intense focus, and absolute stability. Every single incision and manipulation requires a steady hand, supported by tools engineered to exact anatomical specifications. You simply cannot compromise when selecting gear for these intricate procedures, as the structural integrity of your tools directly influences your surgical success.
Grey Medical and Your Surgical Success
The Role of High-Grade Materials
Superior manufacturing always begins with the meticulous selection of premium raw materials. Medical-grade stainless steel and robust titanium form the absolute foundation of our advanced manufacturing process, ensuring maximum durability. These advanced materials easily withstand rigorous instrument sterilization trays cycles without losing their structural integrity, shape, or razor-sharp edges. When you invest in this premium equipment, you secure a reliable asset that maintains its essential calibration over thousands of vital procedures.
Ergonomics in the Operating Theatre
Surgeon fatigue remains a significant concern during lengthy, highly complex eye operations. Prolonged procedures require intense concentration and awkward hand positioning that can easily lead to physical strain or tremors. We deliberately design our ophthalmic surgical instruments with advanced ergonomic principles to actively reduce hand fatigue and improve tactile feedback. This thoughtful design approach ensures that you maintain a comfortable, secure grip, allowing you to focus entirely on the patient.
Maintaining Rigorous Quality Control
Every single tool must pass a series of stringent quality assurance tests before it ever reaches a surgical tray. Our dedicated inspection teams examine each piece under high magnification to verify flawless edges, perfect alignment, and incredibly smooth mechanisms. We leave absolutely nothing to chance, knowing that these highly specialized ophthalmic surgical instruments will touch the most sensitive tissues in the human body. This strict commitment to excellence guarantees that every piece of equipment functions exactly as intended straight out of the packaging.
Sterilization and Longevity
Effective infection control requires equipment that can survive the harshest cleaning protocols without degrading. Our tools feature seamless joints and non-porous surfaces that prevent biological matter from accumulating during operations. You can confidently subject these tools to high-temperature autoclaving, knowing they will emerge completely sterile and ready for immediate use. This remarkable resilience significantly extends the lifespan of your inventory, providing excellent long-term value for your entire medical facility.
Advancing Surgical Outcomes Through Innovation
As medical techniques rapidly evolve, the gear required to perform them must also advance accordingly. We continuously collaborate with leading global surgeons to refine our designs and develop completely new solutions for emerging surgical methods. By staying at the very forefront of medical technology, we actively help you push the boundaries of what is currently possible in eye surgery. Your remarkable expertise combined with our innovative engineering creates a powerful synergy that elevates the global standard of patient care.
Supporting Medical Procurement Professionals
Medical procurement professionals face the daunting task of balancing strict hospital budgets with the need for uncompromised clinical quality. We streamline this challenging process by offering transparent pricing, reliable supply chains, and exceptional customer support for your purchasing department. Your facility requires a dependable partner who delivers crucial orders on time, every single time, without ever sacrificing manufacturing standards. We provide that essential stability, ensuring your surgical teams never experience disruptive equipment shortages.
Elevating Patient Care Standards
The ultimate goal of any medical intervention is to restore health and drastically improve the patient’s quality of life. High-quality tools directly contribute to this goal by enabling smaller incisions, reducing tissue trauma, and promoting significantly faster recovery times. When your surgical team operates with complete confidence in their gear, procedures flow smoothly, and patient safety increases dramatically. We take immense pride in knowing our tools play a small but vital role in preserving and restoring human sight.
Training and Familiarization Protocols
Integrating new equipment into your existing hospital workflows requires clear communication and proper staff orientation. We actively support medical teams by providing detailed handling guidelines and comprehensive care instructions for all our products. Your surgical technicians and nursing staff will quickly master the proper preparation and maintenance routines required for these advanced devices. This thorough understanding prevents accidental damage during routine cleaning and ensures every tool remains perfectly calibrated for its next critical deployment.
Securing Your Premium Surgical Equipment
Equipping your medical facility with reliable tools represents a highly critical investment in your daily practice and your patients. We always stand ready to supply your dedicated team with the finest gear available on the current market. By actively choosing our expertly crafted ophthalmic surgical instruments, you align your practice with decades of proven medical excellence. Contact Grey Medical today to safely upgrade your surgical trays and experience the profound difference that true precision engineering makes in your operating theatre.
Development
KAT6B, MAFB, MAT2A: Precision Trio Transforming Cancer
In the high-speed world of modern molecular biology, we are moving past the simple era of just identifying genetic mutations. Scientists are no longer satisfied with knowing a gene is “broken.” Instead, the focus has shifted toward the complex interplay between epigenetics—the system that turns genes on or off—and metabolic reprogramming, which is how cancer cells fuel their relentless growth. At the center of this scientific revolution are three specific genes: KAT6B, MAFB, and MAT2A. These three players are rapidly becoming known as the “Precision Trio” of oncology and developmental biology.
By understanding how these genes interact, researchers are finding new ways to dismantle tumors from the inside out. They are moving away from broad, toxic treatments and toward therapies that target the very logic of a cancer cell.
KAT6B: The Master Architect of Chromatin
Think of KAT6B (Lysine Acetyltransferase 6B) as the master architect of your cellular blueprint. It doesn’t just hold information; it actively manages it. KAT6B functions as a histone acetyltransferase. This means it adds chemical “tags” to the proteins (histones) around which our DNA is tightly wrapped.
When KAT6B adds these tags, it causes the DNA to relax, making certain genes accessible for the cell to read. If KAT6B is working correctly, your body develops and functions normally. However, if this architect makes a mistake, the results are often catastrophic.
The Link to Cancer and Development
Historically, doctors associated KAT6B mostly with rare developmental disorders that affect the skeleton and brain. However, in 2026, it has become a major target in cancer research. Disruptions in KAT6B are frequently found in Acute Myeloid Leukemia (AML) and several types of solid tumors. When the architect fails, the cell loses its structural and genetic identity. This failure leads to the chaotic, uncontrolled cell division that we define as cancer. By targeting KAT6B, scientists hope to “re-structure” the cell and restore its original, healthy blueprints.
MAFB: The Foreman of Cellular Identity
If KAT6B is the architect, MAFB (MAF BZIP Transcription Factor B) is the foreman on the construction site. While the architect draws the plans, the foreman ensures the workers become what they are supposed to be. MAFB is a transcription factor, a protein that tells a cell its specific identity. It is particularly vital in the development of blood cells and the creation of macrophages, which are the “vacuum cleaners” of the immune system.
A Double-Edged Sword in Oncology
In the medical world, MAFB is a complex character. On one hand, we need it for a healthy immune system. On the other hand, when MAFB is overexpressed—meaning there is too much of it—it becomes a hallmark of multiple myeloma and other blood cancers.
Researchers are now exploring “differentiation therapy” using MAFB. Instead of trying to kill the cancer cells with poison, they are trying to use MAFB to “order” the cancer cells to grow up. If a cancer cell can be forced to mature into a functional, harmless white blood cell, it stops dividing. This approach represents a shift from destroying cells to re-programming them back into a state of health.
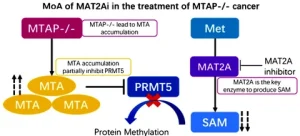
MAT2A: The Powerhouse Supplier of Methylation
The third member of the trio is MAT2A (Methionine Adenosyltransferase 2A). This gene is the critical bridge between what a cell “eats” and how its genes “behave.” MAT2A is responsible for producing S-adenosylmethionine (SAM). SAM is the universal donor for a process called methylation.
Methylation is essentially a “silencing” mechanism. When a methyl group is added to DNA, it usually turns a gene off. For a cancer cell to stay aggressive and immortal, it needs a constant, high-volume supply of SAM. Because of this, many cancer cells become “addicted” to MAT2A.
The Strategy of Synthetic Lethality
One of the biggest breakthroughs in 2026 involves a concept called synthetic lethality. Scientists have discovered that certain tumors (specifically those lacking the MTAP gene) are extremely vulnerable if MAT2A is blocked. By inhibiting MAT2A, we can effectively starve the cancer of the chemical tools it needs to keep its “off-switches” working. Without MAT2A, the cancer cell’s epigenetic logic collapses, leading to cell death while leaving healthy cells relatively unharmed.
The Synergetic Future: A Three-Pronged Attack
The real power of the Precision Trio lies in their synergy. We are no longer looking at acetylation (KAT6B), transcription control (MAFB), and methylation metabolism (MAT2A) as isolated events. Instead, we see them as a single, integrated machinery that drives malignancy.
Why This Matters for Precision Medicine
In the past, doctors treated cancer based on where it was found in the body—lung cancer, breast cancer, or bone cancer. Today, we treat cancer based on its genetic logic.
-
The Architect (KAT6B): Controlling access to the DNA.
-
The Foreman (MAFB): Controlling the cell’s identity.
-
The Supplier (MAT2A): Providing the raw materials for gene silencing.
Pharmaceutical companies are currently developing small-molecule inhibitors that target these three proteins. By attacking the architect, the foreman, and the supplier at the same time, we can dismantle the tumor’s infrastructure. This multi-pronged attack makes it much harder for cancer to develop resistance to treatment.
Improving Readability and Research Access
For scientists in the lab, time is the most valuable resource. To push these discoveries from the bench to the clinic, researchers need high-affinity antibodies and highly specific inhibitors. Fortunately, advanced digital tools now allow scientists to search for these specific gene symbols and immediately access the reagents they need. This digital integration is accelerating the pace of discovery, ensuring that the “Precision Trio” moves into human clinical trials faster than any previous generation of cancer drugs.
Conclusion: A New Era of Hope
The study of KAT6B, MAFB, and MAT2A is a testament to the incredible progress we have made in decoding the human genome. We are finally moving away from the “carpet bombing” approach of traditional chemotherapy. Instead, we are using “surgical strikes” against the proteins that define the cancer’s existence.
As we continue to refine these targeted therapies, the dream of a less toxic, more effective cancer treatment is becoming a reality. By leveraging the combined power of epigenetics and metabolism, we are not just fighting cancer; we are outsmarting it. The Precision Trio is leading the way toward a future where a cancer diagnosis is no longer a sentence, but a solvable genetic puzzle.
-
Business3 years ago
Cybersecurity Consulting Company SequelNet Provides Critical IT Support Services to Medical Billing Firm, Medical Optimum
-
Business3 years ago
Team Communication Software Transforms Operations at Finance Innovate
-
Business3 years ago
Project Management Tool Transforms Long Island Business
-
Business3 years ago
How Alleviate Poverty Utilized IPPBX’s All-in-One Solution to Transform Lives in New York City
-
health3 years ago
Breast Cancer: The Imperative Role of Mammograms in Screening and Early Detection
-
Sports3 years ago
Unstoppable Collaboration: D.C.’s Citi Open and Silicon Valley Classic Unite to Propel Women’s Tennis to New Heights
-
Art /Entertainment3 years ago
Embracing Renewal: Sizdabedar Celebrations Unite Iranians in New York’s Eisenhower Park
-
Finance3 years ago
The Benefits of Starting a Side Hustle for Financial Freedom